Attempting to explain the Mpemba Effect
Posted by David Zaslavsky on — Edited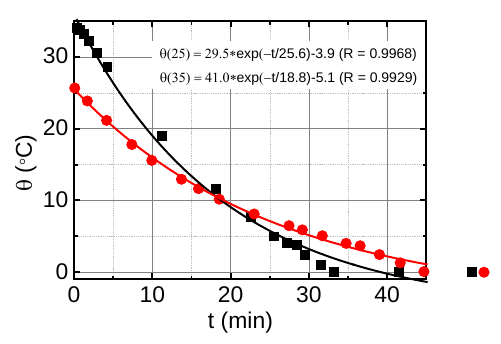
Again entering the realm of interesting physics from last month: a group of scientists proposed a new and quite promising explanation for the Mpemba effect, the observation that warmer water sometimes freezes faster than colder water.
The Definition
Oddly enough, even though the Mpemba effect has been known since ancient times, and its namesake paper was published over half a century ago, physicists can’t even seem to agree on a precise definition of the effect. What we know is that, if you take two containers of water (or, evidently, a water-based mixture like milk) which are identical in every respect except that one is at a higher temperature than the other, and you put them both in a freezer, under some conditions the one at the higher temperature will freeze before the one at the lower temperature. But what conditions are those? And what exactly does it mean to freeze first? Freezing is an extended process, after all; is it talking about when the water reaches zero degrees (Celsius), or when it begins to form ice, or when it is completely converted into ice, or something else?
Part of the contribution of this new paper is a proposal of …
